9.6 - Polar and Non-Polar molecules
We have discussed how bonds can be both polar and nonpolar. These bonds also make the entire molecule polar or nonpolar. Again, it is the polarity that accounts for many of the properties that we see in molecules - most especially, melting and boiling points, as well as solubility.
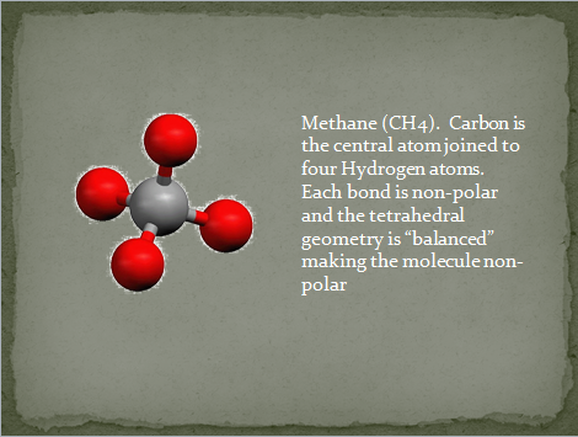
If a molecule is made of nonpolar bonds, like CH4 and most all oils, the entire molecule must also be nonpolar. This means that nonpolar molecules have very little attraction to one another. The molecules don’t collect to each other very well. Unless they are very large individually, nonpolar molecules are gases at room temperature or at least evaporate very easily.
If a molecule is made of nonpolar bonds, like CH4 and most all oils, the entire molecule must also be nonpolar. This means that nonpolar molecules have very little attraction to one another. The molecules don’t collect to each other very well. Unless they are very large individually, nonpolar molecules are gases at room temperature or at least evaporate very easily.
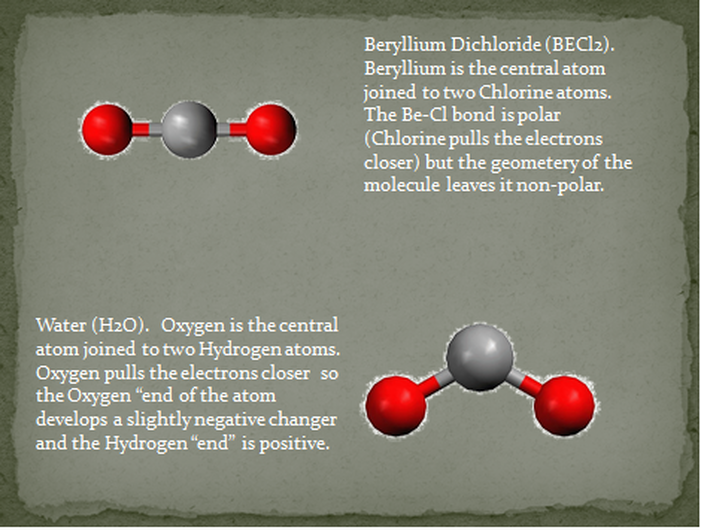
We can think of ionic bonds as being the most polar a bond can be. There is nothing more polar than the ends being different by a whole, complete electrical charge. If a molecule is made of polar bonds, it may be polar or nonpolar. It all depends on the geometry. If the pull from each of the bonds cancels each other out, like forces that are equal in magnitude but opposite in direction, the molecule will be nonpolar. Examples of this are BeCl2, AlCl3 and CF4. Although the bonds are polar, the entire molecule does not have one side that is more or less negative than another. The molecule is nonpolar and so they have little attraction to each other. If the pull from the polar bonds in a molecule do not cancel each other out, the molecule is polar. Examples are NaCl, H2O, and NH3. The shape of the molecule makes one side of it more negative than another- the molecule is polar. Polar molecules do attract each other. They are normally found as liquids and solids at room temperature.